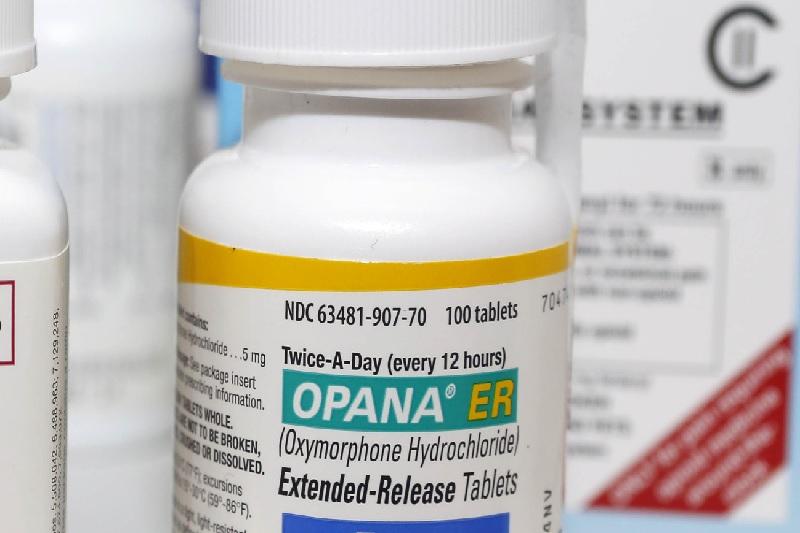
Opana ER was first approved in 2006 for the management of moderate-to-severe pain when a continuous, around-the-clock opioid analgesic is needed for an extended period of time. 
This decision follows a March 2017 FDA advisory committee meeting where a group of independent experts voted 18-8 that the benefits of reformulated Opana ER no longer outweigh its risks. Injection abuse of reformulated Opana ER has been associated with a serious outbreak of HIV and hepatitis C, as well as cases of a serious blood disorder (thrombotic microangiopathy). The FDA’s decision is based on a review of all available postmarketing data, which demonstrated a significant shift in the route of abuse of Opana ER from nasal to injection following the product’s reformulation. 
“We will continue to take regulatory steps when we see situations where an opioid product’s risks outweigh its benefits, not only for its intended patient population but also in regard to its potential for misuse and abuse.” “We are facing an opioid epidemic – a public health crisis, and we must take all necessary steps to reduce the scope of opioid misuse and abuse,” said FDA Commissioner Scott Gottlieb, M.D. This is the first time the agency has taken steps to remove a currently marketed opioid pain medication from sale due to the public health consequences of abuse. After careful consideration, the agency is seeking removal based on its concern that the benefits of the drug may no longer outweigh its risks. Food and Drug Administration requested that Endo Pharmaceuticals remove its opioid pain medication, reformulated Opana ER (oxymorphone hydrochloride), from the market.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
